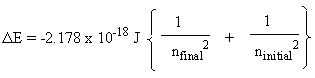How to calculate frequency and wavelength of the radiation emitted from a hydrogen atom when the electron jumps from 3rd level to ground state - Quora

Using the Rydberg formula, calculate the wavelength of the first four spectral lines in the Lyma... - YouTube

How to Calculate the Photon Frequency Absorbed or Emitted by an Electron in a Hydrogen Atom | Physics | Study.com





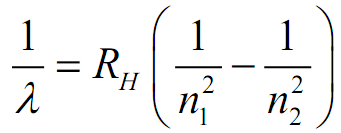







:max_bytes(150000):strip_icc()/what-is-the-rydberg-formula-604285_final-251d1441e24e44c88aab687409554ed4.png)